Metabolic diseases have become a global health crisis, affecting millions of people worldwide. Conditions like obesity, type 2 diabetes (T2D), and renal and cardiovascular diseases are rising, and their impact on individuals and society is substantial1. While genetics, lifestyle, and dietary choices play significant roles in developing metabolic diseases, a growing body of research points to a common underlying factor: inflammation. In this blog, we will explore the inflammation basis of metabolic disorders and understand how chronic low-grade inflammation can silently contribute to their development.
The Interplay of Inflammation and Metabolic Disease: Unravelling a Complex Connection
Inflammation is a complex, highly regulated biological response that helps the body defend itself against harmful stimuli, pathogens or damage. When the immune system detects a threat, it releases soluble mediators and activates immune cells to eliminate the danger and promote tissue repair. This acute, short-term inflammation is a vital part of the body’s immune response and is necessary for healing and recovery from injuries and infections.
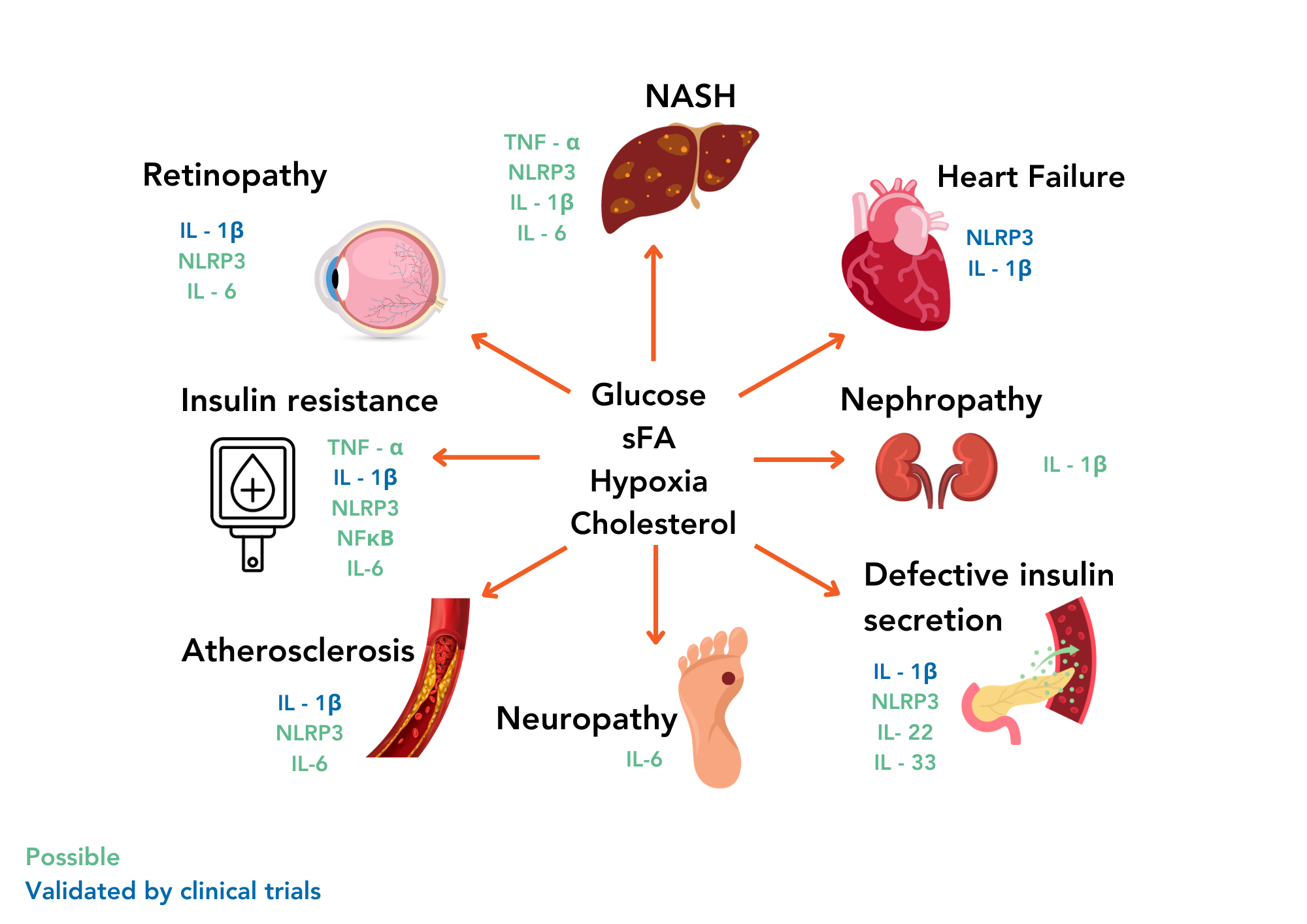
However, chronic, low-grade inflammation is different; it is a persistent, non-resolving, low-level state of inflammation that can have irreversible detrimental effects over time. Such “sterile” inflammation develops without external pathogens and responds to perceived damage due to excess fat and cholesterol, hypertension, stress and ageing2. This type of inflammation is not the typical defence mechanism but an ongoing state of heightened immune activity. Nowadays, chronic low-grade inflammation in metabolic organisms and tissues, often referred to as “metabolic inflammation”, “meta-inflammation” or “metaflammation,” is recognized as a key driver of the development and progression of various metabolic diseases and can be confirmed by increased levels of c-reactive protein (CRP), a general biomarker of systemic inflammation.
Indeed, specific inflammatory signals linked to T2D include adipocyte cytokine production, increased proinflammatory M2 macrophages and the loss of immune-suppressing regulatory T cells. Such changes are believed to be significant contributors to the development and progression of these metabolic conditions. Meta-inflammation can disrupt normal metabolic processes, interfere with regulating blood sugar and lipids, and lead to insulin resistance, a hallmark of T2D.
Fighting Meta-inflammation: Pharmacological Perspectives3
Pharmacological strategies for addressing meta-inflammation involve using medications to prevent or block chronic low-grade inflammation. While these medications are not a single, standalone solution and should be complemented with lifestyle modifications and anti-obesity/T2D therapies, they can be valuable components of a comprehensive treatment management plan.
Here are some potential and current pharmacological strategies for addressing meta-inflammation and metabolic disease:
- General anti-inflammatories4
- Nonsteroidal anti-inflammatory Drugs (e.g., ibuprofen)
- Specific anti-inflammatory drugs
- Anti-cytokine biologics (e.g., anti-IL-1b canakinumab)
- Anti-diabetic medications
- Metformin – is also known to exhibit direct anti-inflammatory effects5.
- Statins – cholesterol-lowering drugs (e.g., atorvastatin)
- Anti-obesity/T2D medications (e.g., the GLP1 agonist, Semaglutide is known to suppress inflammation and CRP levels in obese individuals)6,7
Breaking the Cycle: Is there a best therapeutic approach?
Metabolic diseases and inflammation share a complex interplay, forming a relentless positive feedback loop. As inflammation intensifies, it exacerbates insulin resistance and other metabolic anomalies, further fomenting inflammation. This intricate cycle proves challenging to disrupt, yet breaking it represents a pivotal step in stopping and preventing metabolic diseases.
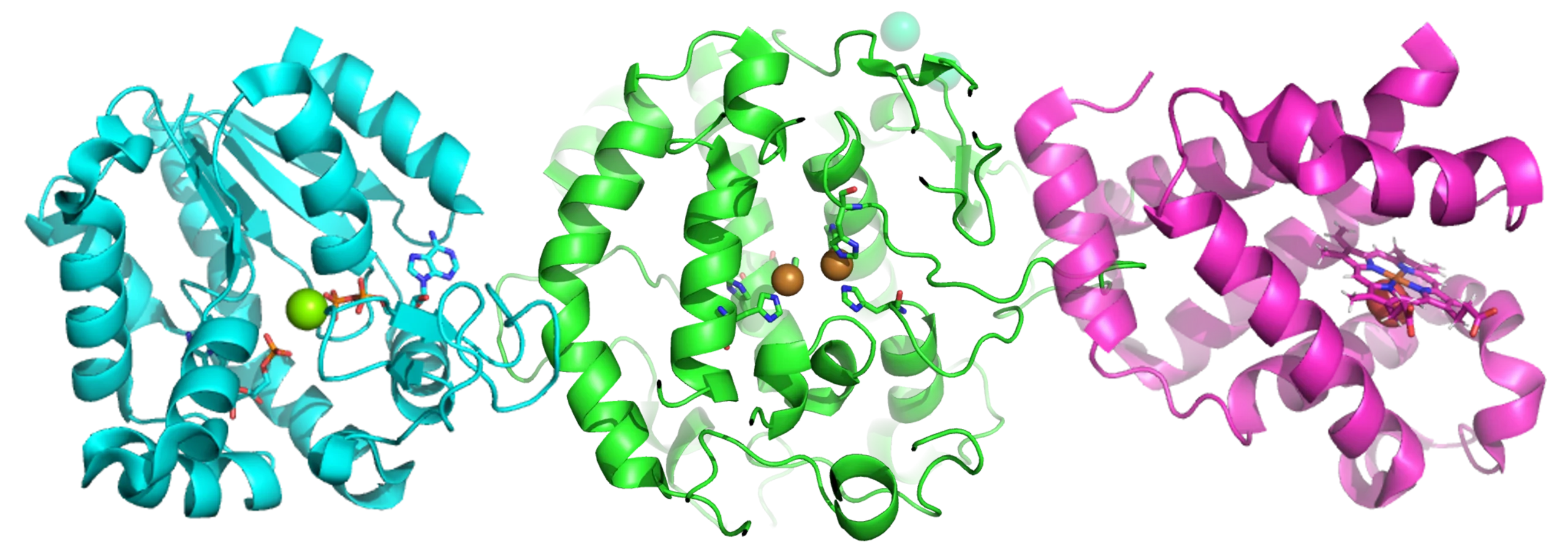
Among the myriad anti-inflammatory strategies under investigation, IRAK4, a crucial kinase in regulating IL-1b production, has garnered attention in the context of metabolic diseases8. Emerging research suggests IRAK4’s potential involvement in the inflammatory milieu of fatty tissue, thereby positioning it as a prospective therapeutic target. Inhibition of IRAK4 activity holds theoretical promise in alleviating the inflammatory burden within tissues like adipose, potentially ameliorating overall metabolic health.
However, it is essential to emphasize that pharmacological interventions for metabolic inflammation mandate individualized consideration, contingent on the specific metabolic ailment, its gravity, and potential adverse effects. Notably, they should be accompanied by persistent lifestyle modifications, incorporating a prudent diet and regular physical activity, as these remain indispensable elements in the comprehensive management of metabolic diseases and the reduction of meta-inflammation.
Conclusion
In combating metabolic diseases, it is essential to recognize the inflammation basis as a significant and often silent player. As our understanding of the intricate interplay between inflammation and metabolic disease deepens, we can develop a more effective therapeutic approach to prevent and manage these conditions.
About the authors
John Unitt – VP of Inflammation and Immunology
Gisele Lincevicius – Associate Principal Scientist, in vivo Pharmacology
Wioletta Pijacka – Associate Director, in vivo Pharmacology
Manoja Brahma Associate Principal Scientist – in vivo Pharmacology
Steve Vickers – Director of in vivo Pharmacology
References
- Sidik, S. M. Diabetes and obesity are rising globally — but some nations are hit harder. Nature (2023) doi:10.1038/d41586-023-00676-z.
- Wu, H. & Ballantyne, C. M. Metabolic Inflammation and Insulin Resistance in Obesity. Circ. Res. 126, 1549–1564 (2020).
- Metaflammation in obesity and its therapeutic targeting | Science Translational Medicine. https://www.science.org/doi/10.1126/scitranslmed.adf9382.
- Li, D., Zhong, J., Zhang, Q. & Zhang, J. Effects of anti-inflammatory therapies on glycemic control in type 2 diabetes mellitus. Front. Immunol. 14, 1125116 (2023).
- Foretz, M., Guigas, B. & Viollet, B. Metformin: update on mechanisms of action and repurposing potential. Nat. Rev. Endocrinol. 1–17 (2023) doi:10.1038/s41574-023-00833-4.
- Verma, S. et al. Effects of once-weekly semaglutide 2.4 mg on C-reactive protein in adults with overweight or obesity (STEP 1, 2, and 3): exploratory analyses of three randomised, double-blind, placebo-controlled, phase 3 trials. eClinicalMedicine 55, (2023).
- Mehdi, S. F. et al. Glucagon-like peptide-1: a multi-faceted anti-inflammatory agent. Front. Immunol. 14, (2023).
- Kondo, M. et al. Therapeutic effects of interleukin-1 receptor-associated kinase 4 inhibitor AS2444697 on diabetic nephropathy in type 2 diabetic mice. Naunyn. Schmiedebergs Arch. Pharmacol. 393, 1197–1209 (2020).