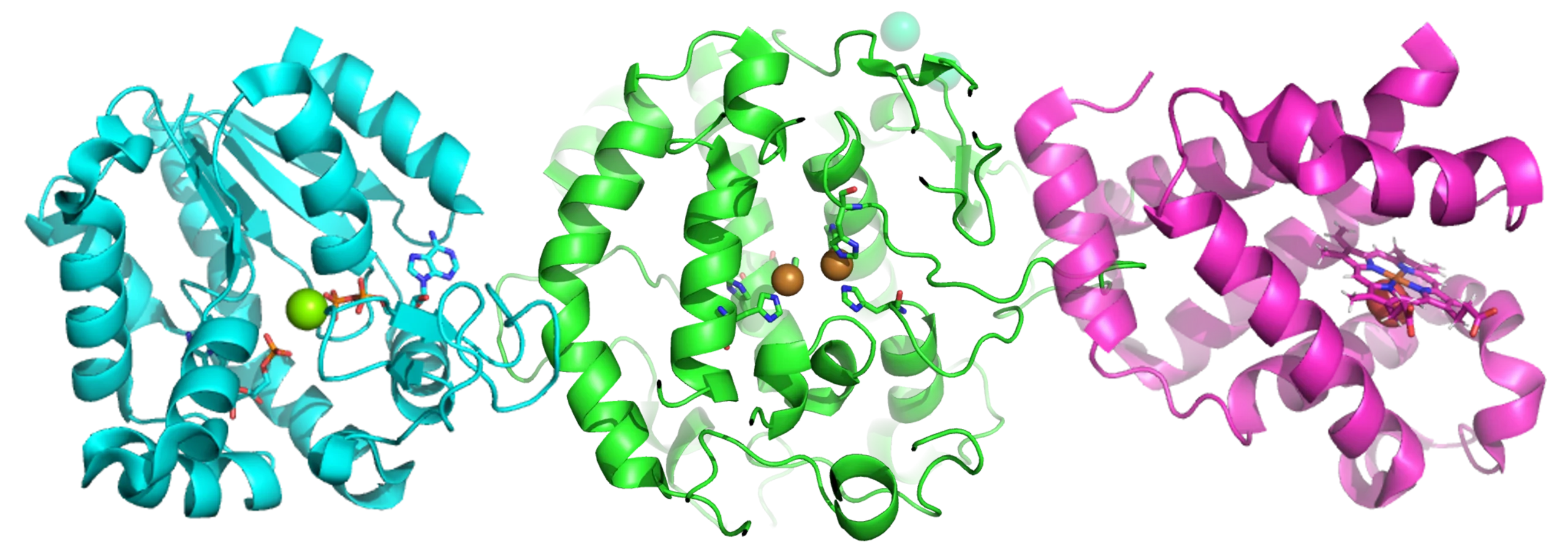
The form, formulation and DMPK parameters of a drug are intricately linked to exposure and therefore to efficacious dose. The solid form of a compound impacts many variables including solubility, bioavailability, tabletability, and physical/chemical stability of the drug. The extent of absorption and hepatic clearance impact oral bioavailability and the rate of absorption relative to distribution and clearance determines half-life. Combined, these parameters determine the exposure for a given dose and dose frequency. Successful drugs need to balance these properties to achieve sufficient exposure for efficacy and to define therapeutic windows in toxicology studies.
The problems customers face with non-linear PK
Form analysis and formulation development need to be brought into the drug discovery process earlier, not left to the point where a candidate has been nominated. Form is often ignored but to do so can cause catastrophic failure down the line as in the case of Ritonavir which was removed from the market when a second more stable polymorph was discovered.
Formulations are considered often at the point where a significant bioavailability issue has been identified or where a compound exhibits non-linear PK and an enabling formulation is required to proceed through pre-clinical toxicology.
Integrating the right teams to overcome these challenges
Form and Formulation should integrate, scientifically and physically, with the DMPK and pharmacology departments. This enables clients to harness the know-how in DMPK to understand PK/PD relationships and calculate the target exposure which drives the therapeutic effect.
With an understanding of the target exposure for efficacy and pharmacokinetics, we can estimate the target absorbed dose and frequency, which are essential to know ahead of formulation development. We recommend the three teams – form and formulation, DMPK and pharmacology – combining to generate and analyse the performance of the formulations.
This integrated approach provides an upfront de-risking strategy as it is able to determine if there are any intrinsic DMPK liabilities with the compound(s) e.g. high clearance that formulation will not resolve before initiating a formulation project.
Delivering value through the right technology
Being technology agnostic is important when developing a formulation strategy. This allows the most appropriate technology to be applied to the molecule and the client’s requirements, rather than ‘shoe-horning’ a compound into a single formulation methodology.
This approach delivers value to the client by generating the best and financially most appropriate solution for the compound at the stage of development, whether the compound is heading into pre-clinical tox or moving from Phase I to Phase II.; Phase III to a marketed drug.

Karina Wojdat – Scientist 1, who is part of David’s team, pictured setting up the spray drier.
Sygnature’s current offering:
Solid form analysis
Typical projects undertake salt screening, polymorphism screening, crystalline landscape analysis, crystallisation development, and stability studies and utilise the following equipment:
- XRPD
- TGA
- DSC
- DVS
- FT-IR
- Crystal 16
- Morphologi G4
- Zetasizer Ultra
Solution state analysis – supporting both solid form and formulation
- HPLC
- UPLC
- NMR
- SFC prep HPLC
Formulation
Typical projects involve developing pre-clinical formulation to resolve nonlinear PK along with early human formulations – de-risking and testing prior to transfer to GMP manufacturer:
- Liquid formulation – traditional solvent/cosolvent mixtures, SMEDDS, SNEDDS, oil in water liposomes, mixed micelles
- Solids – focused on amorphous solid dispersions, lyophilised powders, spray-dried formulations (aq and organic solvent capabilities) along with Turbula blender
- Dissolution testing with both Pion mDISS and USP-type apparatus
- Stability testing to ICH-like conditions – 2 x Memmert chambers, one at 25°C/60%RH and one at 40°C/75%RH
External technologies
- SC-XRD
- Diamond synchrotron for I11 (powder) and I19 (small molecule)
- ss-NMR
- High-filed (800MHz) cryoprobe-based NMR
- Ultra-high-resolution MS including IFS
- SEM including EDX analysis
Key takeaways
When developing a formulation aiming to deliver a dose which has a therapeutic effect, if you do not understand the form of the drug then you are proceeding with inherent risks in your project. Three important points to keep in mind are:
- The bioavailability, of a compound is the fraction of dosed drug which enters the circulation system.
- The form, or forms, a molecule can take are intrinsically linked to its bioavailability.
- If bioavailability is low due to poor absorption (rather than high hepatic clearance) then form and formulation studies have the potential to increase exposure
About Sygnature Discovery’s Form and Formulation Services team
 Sygnature Discovery’s Form and Formulation department was built to react in a rapid nimble manner at the R&D phase to develop formulations. This reduces the cost for clients as the R&D phase can be conducted in a non-GLP environment which develops methods for direct tech transfer into a GLP CDMO.
Sygnature Discovery’s Form and Formulation department was built to react in a rapid nimble manner at the R&D phase to develop formulations. This reduces the cost for clients as the R&D phase can be conducted in a non-GLP environment which develops methods for direct tech transfer into a GLP CDMO.
Working in the non-GLP environment avoids delays that may be experienced with GLP CDMO’s. All projects have equal priority as there is no internal competition with larger GMP manufacturing slots.
David Pearson was brought into Sygnature Discovery to build and lead the form and formulation team.
‘Setting up a new function but within a hugely successful organisation has challenges, but they were alleviated by working closely with world class teams in HR for talent acquisition, facilities for the new lab, IT support for all the instruments and finance who were happy to keep saying yes every time I asked. We were up and running generating data for clients within a few weeks – result!’

– David Pearson, Director and Form and Formulation at Sygnature Discovery.
To learn more about how the Form and Formulation Services team at Sygnature Discovery can help you with your next project, get in touch using the contact form below.



















 Sygnature Discovery’s
Sygnature Discovery’s