The aim of the lead optimisation stage is to move potent and safe drug candidates through to evaluation ahead of clinical trials.
It’s a crucial step in a drug’s development.
The process focuses on honing in on those all-important drug features to improve the biological activity, target selectivity and toxicity profiles of lead drug candidates while maintaining both good laboratory practice safety and chemistry, manufacturing, and controls studies.
No mean feat, then.
In fact, drug lead optimisation requires a wide range of in-depth knowledge about how your drug leads can have a beneficial effect on disease. To overcome challenges in the translational pathway to get drug leads to the clinic, you need substantial intellectual input from scientists across a breadth of disciplines and backgrounds.
The most successful lead optimisation projects are comprised of truly integrated teams who accelerate drug development in optimised work environments. But why, and what’s involved?

Iterate to optimise
Like in the hit-to-lead phase, the design, make, test, analyse (DMTA) cycle is a fundamental strategy in lead optimisation. Iterations of this cycle enable chemists to tweak structural components of the compounds that can affect potency and toxicity, or improve other desired attributes like affinity, selectivity, and synthesisability. It’s during this process that lead candidates are optimised, and hopefully boiled down to one or two solid candidates.
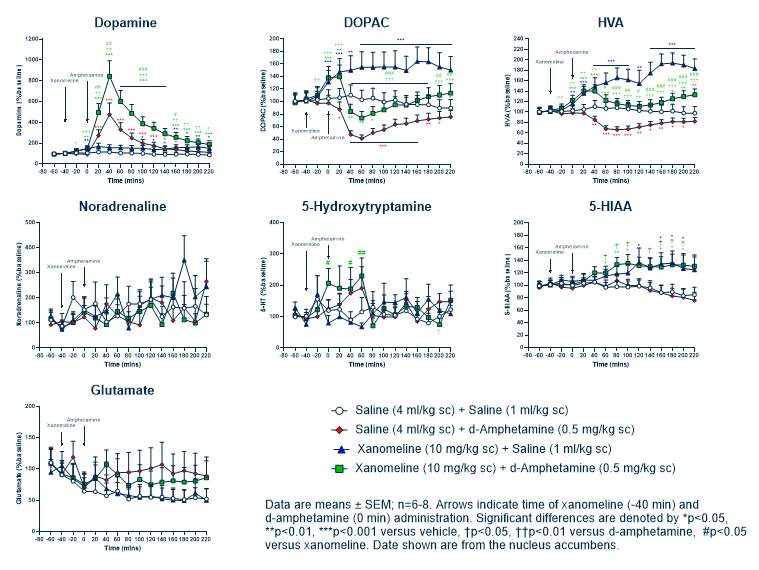
In the lead optimisation stage, fewer compounds are profiled with more assays. Expanding the in vitro and in vivo testing parameters addresses the key challenges of improving the absorption, distribution, metabolism, excretion, and toxicity profile of a selected candidate via ADMET/DMPK studies.
Using a range of assays empowers you to answer the crucial questions that will affect the future viability of a project; namely efficacy, selectivity, formulation, scale-up and toxicity. This stage sees a shift from more in vitro based assays to in vivo animal models of disease. The goal here is to increase the confidence that the drug candidates have fundamental beneficial effects on the human disease state.
To ensure the results from in vivo studies translate to the clinic, it’s important for teams to consider which animal species and models are used to predict human outcomes and better validate their drug.
Flexible assay cascades
Having flexible assay cascades rather than formulaic protocols is an important aspect of lead optimisation.
Taking a flexible approach enables you to focus on the challenges for that particular lead series and tackle any specific organ toxicity issues head on. Using a range of in vitro models, such as hepatocyte models, for example, can reveal if there is likely to be drug-induced liver injury; a significant cause of new drug failures.
Once you’re confident in your lead compound, the target-related biology can be elucidated by testing the compound in a broader panel of mechanistic and functional cell systems.
By doing so, you can be assured that modulating the target does indeed have a beneficial effect on treating the disease; strengthening the translational pathway to the clinic.
Integrated approaches to lead optimisation
Lead optimisation requires substantial input from scientists across a range of different disciplines and backgrounds.
The emphasis on this stage is the quality of your assay cascades, drug structure adaptations, and safety and efficacy profiles that will develop a lead candidate into a specialised drug that will drive beneficial effects in clinical trials.
That’s why true integration of experts and their knowledge is so crucial. Interdisciplinary communication at every level encourages innovation and project excellence that can accelerate lead optimisation.
Integration enables effective team-based decision making in the DMTA cycle that can lead to effective improvement of a drug candidate.
Simply put, integrating a broader range of expertise into lead optimisation boosts your chances.