Sygnature Discovery has established an in vivo LPS model of inflammation which can help understand life-limiting neuroinflammatory conditions like Parkinson’s disease, multiple sclerosis, Alzheimer’s disease, and traumatic brain injuries and identify new drug targets.
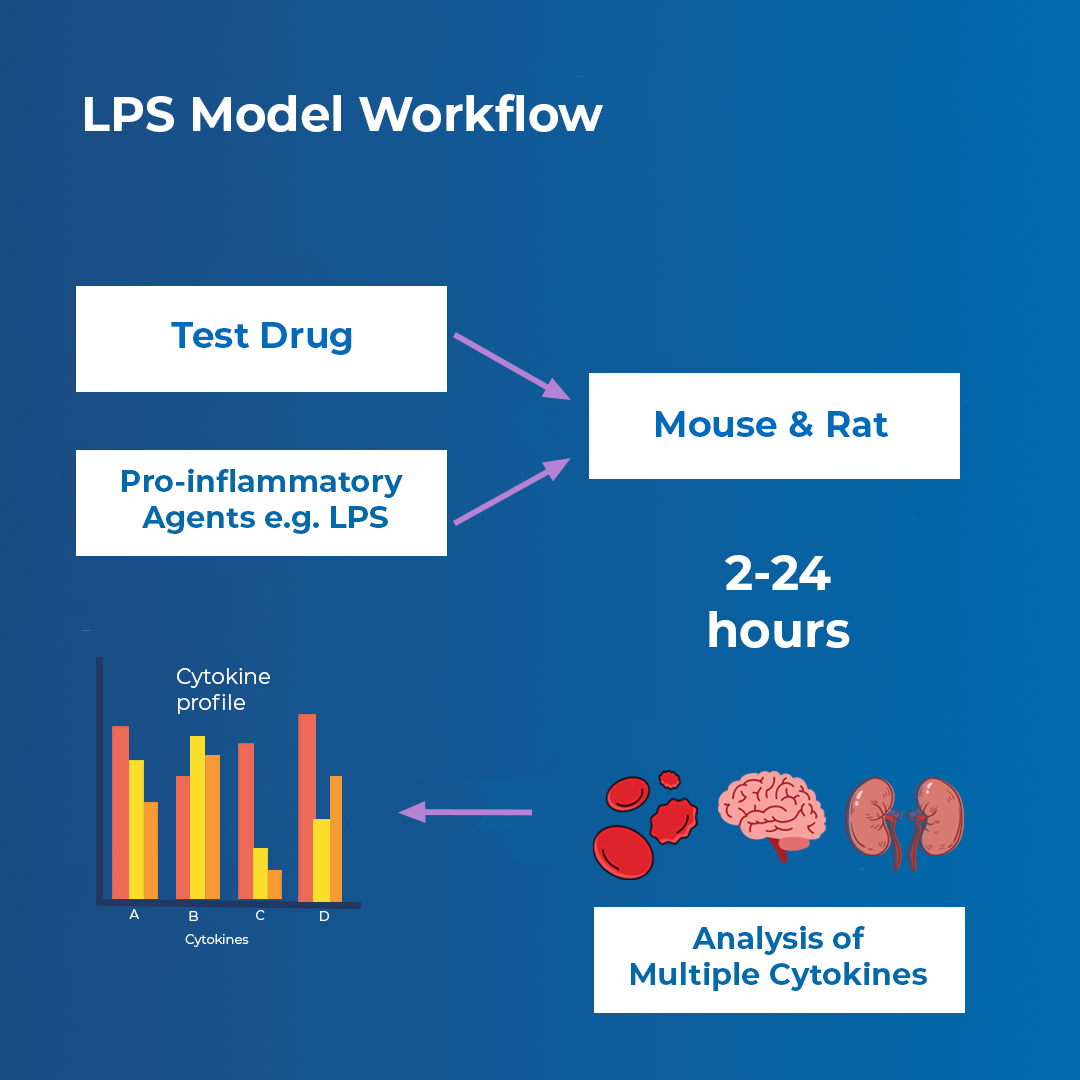 The LPS in vivo model supports early-stage anti-inflammatory drug discovery in neuroinflammation, kidney, and systemic inflammation.
The LPS in vivo model supports early-stage anti-inflammatory drug discovery in neuroinflammation, kidney, and systemic inflammation.
The inclusion of kidney inflammation and neuroinflammation studies in Sygnature Discovery’s LPS model sets it apart from conventional models, providing valuable insights into the complex interplay of inflammation in different organs. This unique capability enables researchers to identify new drug mechanisms, evaluate drug efficacy, and enhance our understanding of kidney and neuroinflammatory diseases, driving the downstream development of targeted therapeutic interventions for patients with unmet needs.
The model also enables an understanding of the pharmacology and processes that underpin systemic inflammation, and builds on existing in vitro capabilities within Sygnature Discovery to further strengthen drug target validation and translation from in vitro to in vivo. It measures key pro-inflammatory cytokines in blood and provides a useful model system to quickly evaluate the efficacy of novel anti-inflammatory drugs in early drug discovery.
Commenting on the model’s benefits Dr. John Unitt, Vice President of Inflammation and Immunology at Sygnature Discovery, said:
“An important step in drug discovery is the successful translation of a drug’s effect on in vitro cell function to efficacy in an in vivo disease model. Sygnature’s LPS in vivo model provides that initial step in determining the efficacy of a novel anti-inflammatory. Getting this first in vivo inflammation model off the ground is an exciting start, but it is only the beginning of a pipeline of new models planned to test a broader range of immunomodulatory drugs and mechanisms.”
Dr. Max Mirza, Vice President of Neuroscience at Sygnature Discovery, added:
“Neuroinflammation like systemic and kidney inflammation involves many of the same inflammatory mediators (cytokines, chemokines). Working across therapeutic teams at Sygnature to establish the LPS model and measure these inflammatory mediators in different parts of the body allows our clients to gain a more holistic answer to their scientific questions. Whether that be a better understanding regarding the mechanism of action, the role of a target on inflammatory processes in multiple body compartments simultaneously, or relevance to a drug’s efficacy. This unique approach allows our clients to understand if impacting systemic inflammation can influence neuroinflammation, and whether systemic inflammatory mediators are potential translatable biomarkers of neuroinflammation.”
To learn more, visit the Sygnature Discovery website for details on the LPS model, or meet with John Unitt at the upcoming World Congress Basic & Clinical Conference in Glasgow, July 2-7 where his team will be presenting on the topic.