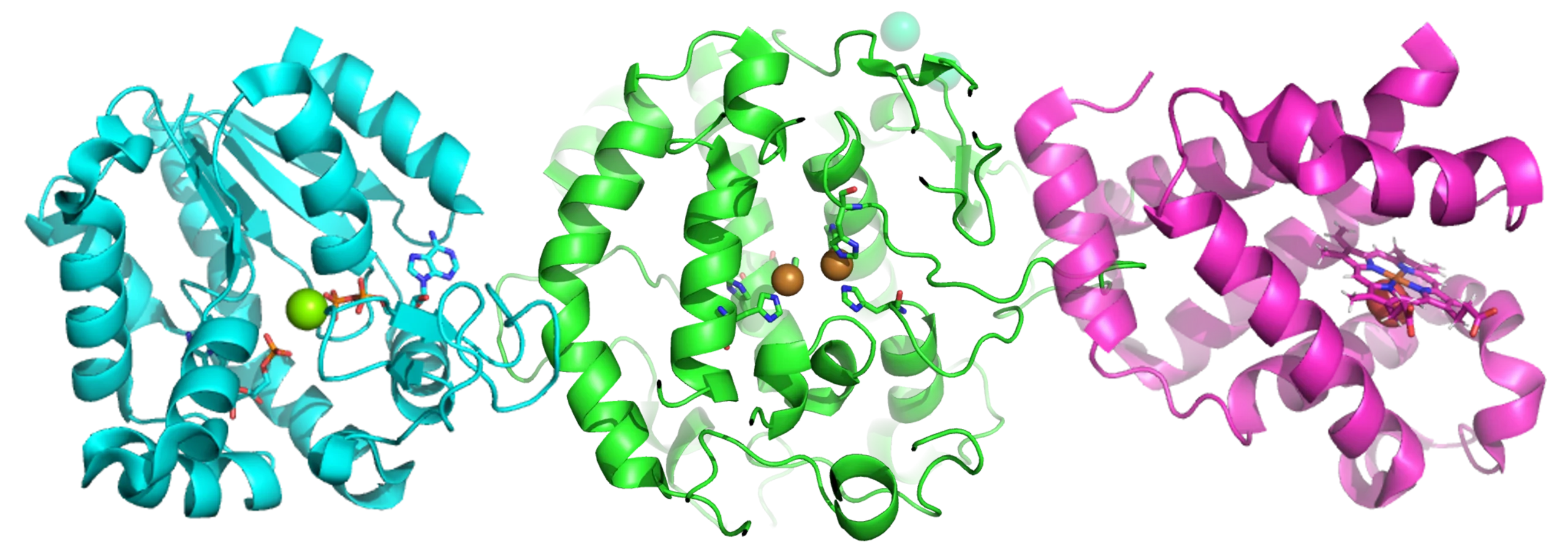
As we await the FDAs decision on Lykos Therapeutics’ NDA for MDMA-assisted therapy for patients with PTSD, companies focussed on psychedelics and psychoplastogen’s are exploring multiple indications where these drugs might provide therapeutic benefit. Recent clinical and preclinical focus from such companies is pushing beyond treatment-resistant depression and PTSD, including ADHD, eating disorders, anxiety, substance use disorder, alcohol dependence, pain, post-partum depression, and adjustment disorder in cancer.
Some companies are focusing on agents that give you the psychedelic experience, as this is thought to be part and parcel of the therapeutic response. However, limiting the length of the psychedelic trip is a key focus for companies in this space, requiring deep knowledge and experience of pro-drug approaches, ADME, and formulation. Others believe you can separate the therapeutic benefit, driven by neuroplastic changes in the brain, from the psychedelic experience. This approach would lead to better-tolerated drugs with a more traditional commercial path forward. However, measuring and defining neuroplastic changes in vitro, ex-vivo and in vivo requires a deep repertoire of skill sets, experience, technology, and knowledge of biology.
Partnership deals involving psychedelic drugs saw a 500% growth in total deal volume from 2019 to 2023, with more than 40 partnerships for psychedelic drugs since 2019.
This year (May 2024) Gilgamesh and Abbvie signed an agreement which gave Abbvie an option to Gilgamesh’s neuroplastogen’s. This is interesting, as it appears to be one of the first examples of a big Pharma stepping into the psychedelic arena as relates to modulating the serotonergic system. The focus, however, is on an approach that avoids the psychedelic trip/hallucinations but still leads to therapeutic benefits in depression and anxiety. Another big Pharma, Johnson & Johnson, already have the psychedelic drug Spravato® (esketamine) approved in the US and Europe since 2019 for Treatment-Resistant Depression (in combination with an oral antidepressant). However, this drug targets the NMDA receptor rather than the serotonergic system.
In the same month, Reunion Neurosciences secured over $100M Series A led by Novo Holdings and MPM BioImpact to develop the psychedelic agent REL-104 for post-partum depression. In contrast to the approach from Gilgamesh and Abbvie, the approach here will engender a psychedelic response, albeit one of shorter duration compared to psilocybin. The lead indication, post-partum depression, is also an interesting choice given Sage Therapeutics succeeded with the first regulatory approvals for PPD with brexanolone and zuranolone in 2019 and 2023, respectively. Reunion has plans for indication expansion, including adjustment disorder in cancer.
Regulatory agencies are responding to the innovation and momentum in the psychedelic drug space, with both the FDA and EMA issuing guideline documentation to support clinical trials and development of psychedelic drugs. Health Canada was the first regulatory authority to approve a clinical trial for psilocybin-assisted therapy, in 2020. Oregon was the first US state to legalize psilocybin for therapeutic use in supervised settings in 2020. Similarly, Canada’s Special Access Program in 2022 allowed healthcare practitioners to request psychedelic drugs for patients. These events shape the ecosystem and spur increased interest from investors, including big Pharma.
The outcome of the FDA advisory committee meeting to discuss Lykos Therapeutics MDMA-assisted therapy on June 4th is another important milestone which will be keenly watched by companies and investors focussed on both psychedelic and non-psychedelic psychoplastogen approaches for treating patients.
About the Author

Dr Max Mirza is a neuroscientist who has worked in biotech, and big and midsize pharma for over 25 years. He has run and overseen multiple drug discovery projects from medicinal chemistry to Phase 2a, working with internal teams, CROs, academics, and investors to progress projects using a stage-gated, decision-guided approach.
At the biotech NeuroSearch he was involved in multiple multi-year collaborations with major pharma companies focused on CNS & Pain projects from research to early clinical development.
He co-founded and was CSO of the CNS-focused biotech Saniona, listed on the Stockholm Nasdaq.
At Pfizer, he was part of the external R&D team focused on bolstering the internal pain and neuroscience pipeline and capabilities, working closely with commercial and BD teams, with an emphasis on novel therapeutic modalities.
Prior to joining Sygnature Discovery, Max was at Mundipharma where he was charged with innovating in pain, neuroscience, metabolic disorders, and infectious disease working closely with commercial and reimbursement teams. Max continues to consult for various VCs and investment teams in neuroscience.